
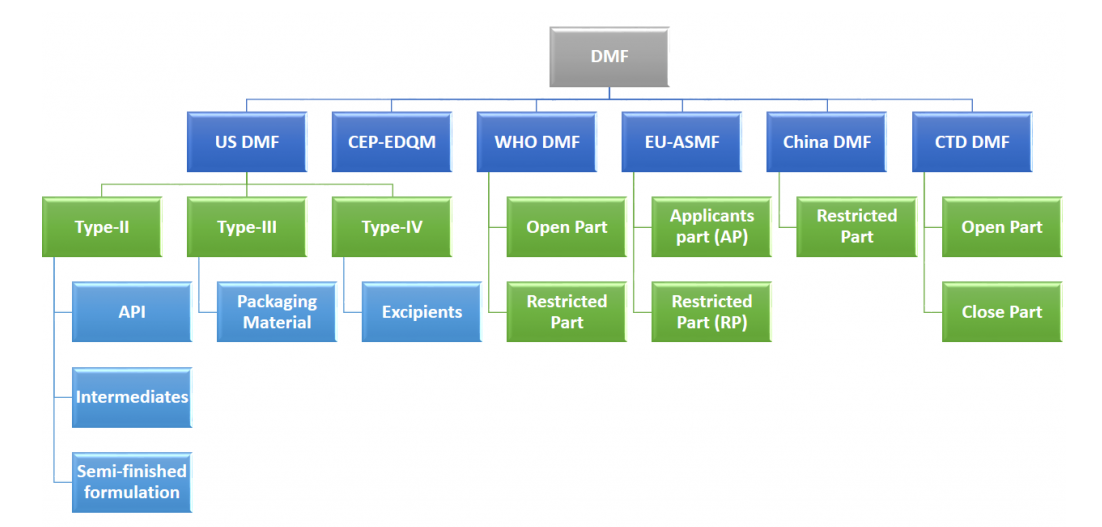
Drug Master File (DMF) is a master file containing complete information on an API/Bulk Drug Substance.It is a submission to FDA covering factual and complete information on its chemistry, stability,purity,impurity profile,packaging and cGMP status of any API. The Main Objective of DMF is to meet regulatory requirements and for Market Authorization (MA).
Our Expertise
Compilation of complete DMF & Submission of the same.
Complete support for preparation of necessary documentation and submission to US FDA along with answers to Post submission queries
Preparing US DMF for Active Substance (Bulk Drug), Excipients, Intermediates, Primary packaging material & submitting the same.
Reactivation of Inactive US DMF, Filling Annual Reports , Submitting LOA letters to clients and FDA.

Our Experience
We have Filled more than 40 DMFs in USA for Type II - Active Ingredient (API) type III - Primary Packaging Material & Type IV - Excipients
Prepared & compiled more than 100 DMFs for Regulated & Semi Regulated Market.
Our services are tailored to meet client’s requirements, and add value to their company by minimizing both time and money spent.
MEDATEQ LABS also perform the following studies, in co-operations to DMF preparation:
Structural Elucidation Studies
Analytical Report (COA)
Analytical Method Validation
Process Validation
Stability data studies
Determination & Validation of Potential Impurities & Solvent Impurities
(All the data supported by necessary Chromatograms & Spectrums)

